BiteSized Immunology: Cells

Dendritic Cells
Dendritic cells (DCs), named for their probing, ‘tree-like’ or dendritic shapes, are responsible for the initiation of adaptive immune responses and hence function as the ‘sentinels’ of the immune system. Paul Langerhans first described DCs in human skin in 1868 but thought they were cutaneous nerve cells. DCs are bone marrow (BM)-derived leukocytes and are the most potent type of antigen-presenting cells. They can also be propagated in vitro from BM and blood using various combinations of growth factors, such as granulocyte macrophage-colony stimulating factor (GM-CSF) and Flt3 ligand. DCs are specialised to capture and process antigens, converting proteins to peptides that are presented on major histocompatibility complex (MHC) molecules recognised by T cells. DCs are heterogeneous, e.g. myeloid and plasmacytoid DCs; although all DCs are capable of antigen uptake, processing and presentation to naive T cells, the DC subtypes have distinct markers and differ in location, migratory pathways, detailed immunological function and dependence on infections or inflammatory stimuli for their generation. During the development of an adaptive immune response, the phenotype and function of DCs play an extremely important role in initiating tolerance, memory, and polarised T-helper 1 (Th1), Th2 and Th17 differentiation.
DCs linking innate and adaptive immunity
Since DCs have numerous cytoplasmic processes, they have a high surface area permitting intimate contact with a large number of surrounding cells, e.g. T cells, natural killer cells, neutrophils, epithelial cells etc. For instance, experimentally, only one mature DC (mDC) is required to stimulate 100–3000 T cells. DC precursors migrate from the BM through the blood stream to almost every non-lymphoid tissue, where they reside in an immature state (iDC), continuously sampling their environment by endocytosis, macropinocytosis, and phagocytosis. They can extend their processes through the tight junctions of epithelia to increase capture of antigens even when there is no overt infection/inflammation. During pathogen invasion, resident iDCs detect intruders via pattern recognition receptor (e.g. TLRs) capture antigens and quickly leave the tissue. They crawl through the cells, cross the endothelium of lymphatic vessels and migrate to the draining lymph nodes (LN) in response to a number of chemokines such as CCL19 and CCL21. During their migration from the peripheral tissues, DCs undergo phenotypical and functional maturation. Most remarkably, they stop capturing antigens while up-regulating the expression of co-stimulatory molecules such as CD80 and CD86 and the chemokine receptor CCR7, and secrete pro-inflammatory cytokines such as TNF-α and IL-12. After reaching the subcapsular sinus of the LN, DCs move to T-cell zones. Here, the interdigitating DCs are actively involved in the presentation of antigens to T cells.
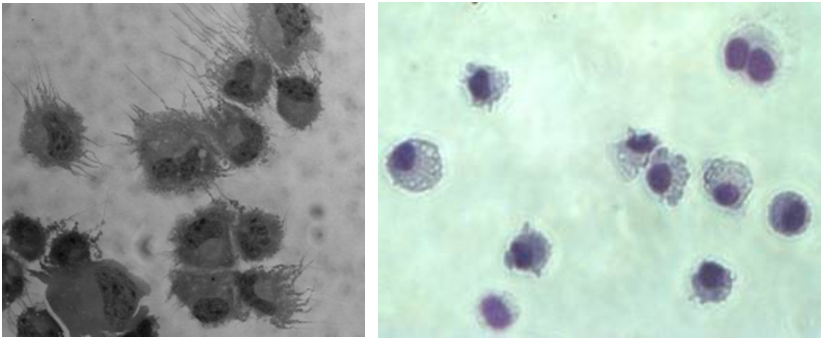
Left: LPS-matured murine BM-derived DCs.
Right: Isolated murine lung CD11c+ and MHCII+ DCs.
DC immunotherapy
Exploiting the immune-regulatory capacities of DCs holds great promise for the treatment of cancer, autoimmune diseases and the prevention of transplant rejection. Manipulation of DCs could turn them into the most effective adjuvant to enhance the host’s immune defences. In the case of cancer, tumours have been shown to suppress DCs by secreting anti-inflammatory cytokines such as IL-10, and therefore conditioning the local DCs to form suppressive T cells. In order to subvert these mechanisms, DCs could be generated ex vivo, loaded with tumor antigens, and re-injected to boost the host’s immunity against the tumour cells. DC vaccines generated in this way are generally safe with minimal side effects, and have proven to be feasible, and effective in some patients. Other strategies exploiting DCs in various disorders have also been described and are being investigated in clinical trials.
© The copyright for this work resides with the BSI
