BiteSized Immunology: Cells

T follicular helper cells
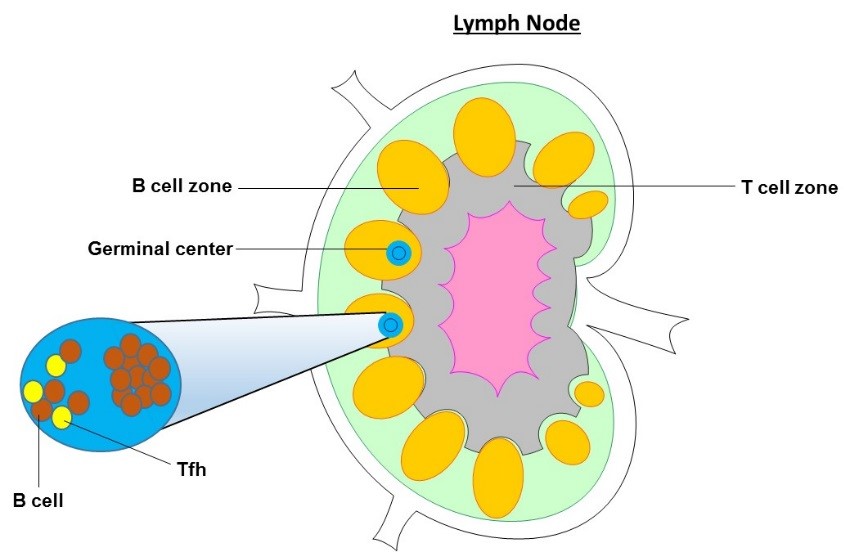
T follicular helper cells (Tfh) are a specialized subset of CD4+ T cells that were first identified in the human tonsil. They play a critical role in protective immunity helping B cells produce antibody against foreign pathogens. Tfh are located in secondary lymphoid organs (SLOs), including the tonsil, spleen and lymph nodes. These organs contain numerous lymphocytes, separated into defined T and B cell zones. Uniquely, Tfh are found in the B cell zone and spend the majority of their time in close interactions with B cells. In addition to SLOs, Tfh can also be identified in the circulation.
Tfh function
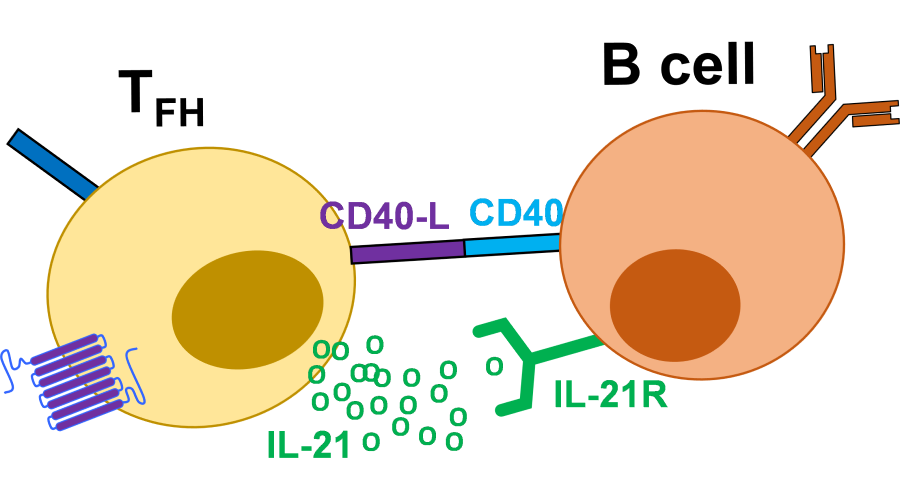
Tfh play an essential role in the formation of germinal centres (GCs), which are distinct structures that form within the B cell zones of SLOs during an ongoing immune response. B cells within GCs are known as GC B cells and undergo rapid proliferation and antibody diversification, allowing the production of many types of antibody, with greater affinity for their targets. GCs are also the site where B cells can differentiate into antibody secreting plasma cells and memory B cells which allow long lasting antibody production. Tfh direct this process by directly providing co-stimulation to the B cells via the co-stimulatory molecule CD40 interacting with CD40-ligand (CD40-L) on the B cell and by producing the cytokine IL-21 which drives B cell proliferation. Additional cytokine production by Tfh is able to determine the type of antibody produced. The relationship between Tfh and GC B cells is positively correlated. In the absence of Tfh, GCs do not form and antibody defects are observed under typical situations.
Tfh differentiation
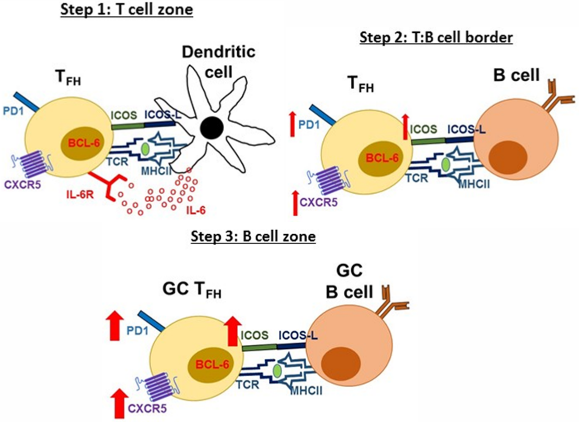
Tfh are defined by expression of the transcription factor, B cell lymphoma 6 (Bcl6) and a number of cell surface markers including CXCR5, PD1 and ICOS. Tfh differentiation is a multi-step process, beginning in the T cell zone of SLOs. Firstly, naïve CD4+ T cells recognize via their T cell receptor (TCR), protein peptides presented on MHC Class II molecules by dendritic cells (DCs). This along with additional stimulation via the co-stimulatory molecule, ICOS-L and IL-6 cytokine production by DCs provides signals to the CD4+ T cell to express Bcl-6 and commit to the Tfh lineage. Developing Tfh express the chemokine receptor CXCR5 and down-regulate the chemokine receptor CCR7, allowing the cells to migrate towards the B cell zone. At the T and B cell zone border, TCR and ICOS-L co-stimulation is handed over to activated B cells allowing developing Tfh to express higher levels of CXCR5, ICOS and PD1 and move to the B cell zone. Eventually, Tfh help form GCs and fully differentiated Tfh within the GC are known as GC Tfh and express the highest level of CXCR5, PD1 and ICOS.
Tfh and disease
Tfh function has been shown to be dysregulated in a number of diseases of excessive or insufficient antibody production. Circulating Tfh numbers have been shown to increase in number in the blood of patients with autoimmune diseases, including systemic lupus erythematosus and rheumatoid arthritis. Patients with common variable immunodeficiency caused by ICOS deficiency have been shown to have severely reduced circulating Tfh and major defects in antibody generation. Lastly, defects in Tfh help to B cells has been observed in HIV infected patients and contributes to the inability of patients to produce effective HIV specific antibodies. Given the contribution of Tfh to a number of human diseases, better understanding of these cells could one day be therapeutically beneficial. Furthermore, the production of long lasting specific antibodies forms the basics of successful vaccination, therefore a great deal of research is being carried out to better understand Tfh to improve vaccine design.
Alternative Tfh
In addition to Tfh expressing the CD4 co receptor, a population of T cells expressing the CD8 co-receptor have been found to express CXCR5, PD1 and Bcl-6. They have been identified in the B cell follicles of mice during both acute and chronic lymphocytic choriomeningitis virus infection. This newly identified population is currently being studied by multiple research groups. It will be interesting to determine their importance to protective immunity.
© The copyright for this work resides with the BSI
This article was updated in 2021
