BiteSized Immunology: Experimental Techniques

Cytokine ELISPOT & FluoroSpot Assays
Principles of the assay
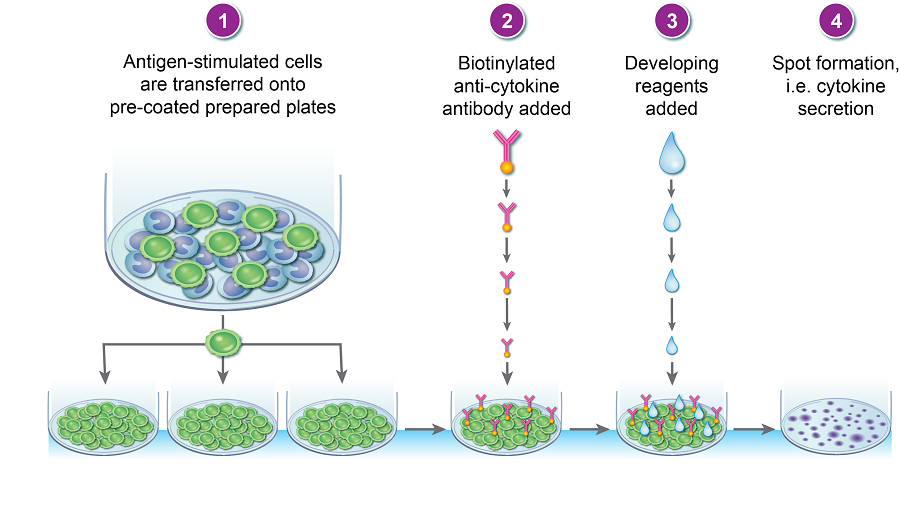
The cytokine ELISPOT is both a quantitative and qualitative assay, it is based on the enzyme-linked immunosorbent technique and is designed to enumerate cytokine-secreting cells; it is extremely sensitive and therefore useful in detecting low frequency cytokine-secreting cells (1/300 000). The cytokine released in response to antigen can be mapped to a single cell cells hence T cell responder frequencies can be calculated. Cells can be stimulated either in the anti-cytokine antibody coated plate (direct assay) or pre-stimulated and then transferred to the pre-coated plate (indirect assay) (Figure 1); once the assay is complete, the plates can be analyzed on a plate reader.
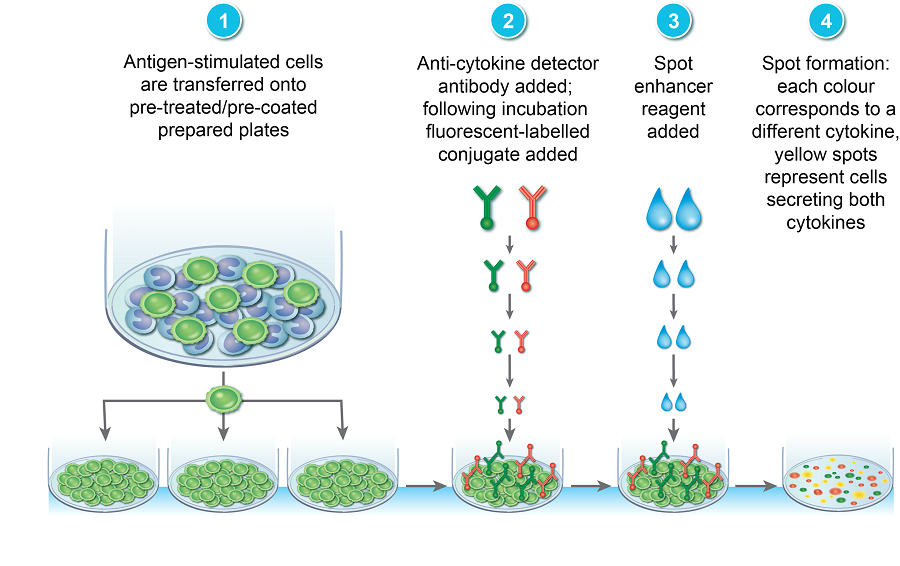
More recently, the assay has been adapted to a FluoroSpot assay which utilizes fluorochrome-conjugated detection antibodies thereby allowing the simultaneous detection of multiple distinct cytokines and subsequent T cell sub-population analysis (Figure 2).
The main application of the ELISPOT/FluoroSpot assay is in monitoring of immune responses in both humans and animals; a modified version is currently used as a diagnostic tool for tuberculosis.
Application of the cytokine ELISPOT assay
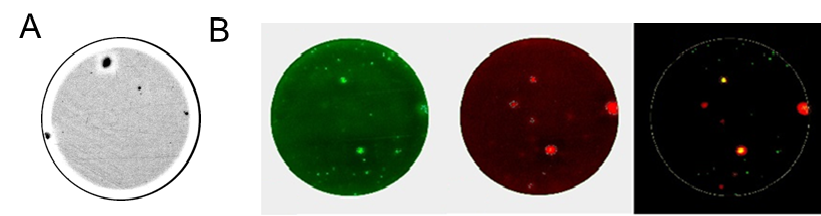
The cytokine ELISPOT has been successfully used across several disciplines of Immunology, including organ-transplantation, cancer research, infectious diseases, vaccine development and autoimmune diseases where the sensitivity of these assays has been shown to be very useful in detecting autoreactive cells which typically occur at low frequencies (Figure 3). Both ELISPOT and FluoroSpot assays are widely used in immuno-monitoring of clinical trials where both quantitative information and T cell phenotype identification at a single cell level is highly informative.
© The copyright for this work resides with the BSI
